Published: Mar 21, 2022
Summary:
Biocon is a global biopharmaceutical company finding affordable ways to treat diabetes, cancer, and autoimmune diseases. Its businesses include Generics, Biosimilars, Research Services, and Novel Biologics. It has 13,500+ employees.
Biocon Biologics, a subsidiary of Biocon, develops high-quality affordable biosimilars.
Biocon Biologics buys Viatris biosimilars assets for $3.33 billion. Viatris will receive $2.33 billion in cash and $1 billion worth of Biocon Biologics shares (12.9% equity stake). Biocon Biologics is valued at $7.75 billion.
Biocon will fund the deal via $800 million of equity infusion and $1.2 billion of debt.
In Nov 2020, Viatris was formed through the merger of Mylan and Upjohn, a division of Pfizer. Its biosimilars business is expected to report revenue of $875 million and EBITDA of $200 million for the year 2022.
Syngene, a subsidiary of Biocon, is a contract research organisation (CRO) from lead generation to clinical supplies. Its clients include Bristol-Myers Squibb, Baxter, Amgen, GSK, Merck KGaA and Herbalife. Biocon holds 70.1% stake in Syngene.
Quick Facts:
- First listed ‘pure play’ bio-pharmaceuticals company in India.
- First company from India to have a biosimilar commercialised in the U.S and in Japan.
- Developed and launched Itolizumab – the world’s first novel anti-CD6 monoclonal antibody.
- One of the largest statins manufacturers in the world, supplying drug substances to over 60 countries.
- One of the largest producers of immunosuppressant APIs globally.
- One of India’s largest biomanufacturing facilities for insulins.
- Asia’s largest integrated insulin manufacturing and R&D facility in Malaysia.
- Among the leading insulins players globally.
- Among Top 10 Global Biotech Employers as ranked by prestigious US-based Science magazine.
- Key Therapeutic Areas: Diabetes, Oncology and Immunology.
- Patents: Small Molecules (299), Biologics (982).
- 25+ cGMP Approvals international regulatory agencies.
- 8 Biologics Approved: 2 Novels and 6 Biosimilars.
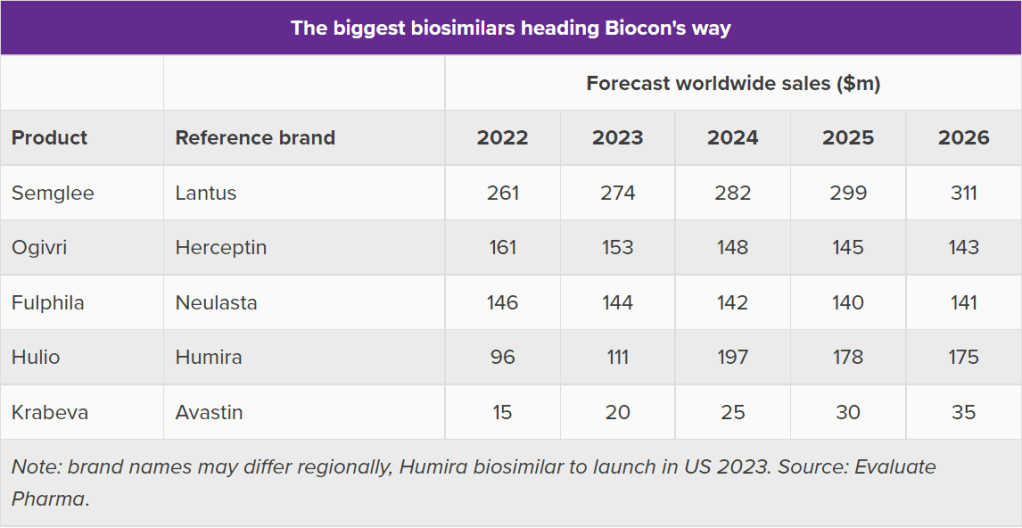
- FDA approved the first biosimilar version of insulin glargine from Viatris and Biocon Biologics. The new treatment, Semglee is a biosimilar version of Sanofi’s insulin Lantus.
- Employee Base: Biocon (25%), Biocon Biologics (35%), Syngene (40%).
Competitors:
- Top 5: Pfizer, Amgen, Biogen, Samsung Bioepis, Novartis.
Revenue:
- Revenue Mix: Generics (31%), Biosimilars (37%), Research Services (29%), Others (3%).
- Revenue Mix: USA (33.2%), India (19.1%), Ireland (18.8%), Rest of the World (28.9%).
- Operating Profit Break-Up: Biosimilars (51.7%), Research Services (33.5%), Generics (14.8%).
Opportunity:
- $75 billion will be the size of biosimilar business globally in next 10 years.
Risks:
- Uncertainty in payoffs from a high R&D driven model in biosimilars.
- Intense competition in the regulated markets.
- Integration with Viatris could be a challenge.
- Expensive valuation as compared to its past valuations.
Negatives:
Biosimilar business is a richly valued in Asia. The Viatris deal is valued at 16.5 times EV/EBITDA compared to the current multiple of 6.5 times. Biocon’s total debt will rise to $1.5 billion.
Management:
- Ms. Kiran Mazumdar-Shaw founded Biocon in 1978
- Ms. Kiran is the Chairperson and MD of Biocon and Biocon Biologics
- Ms. Kiran is an inspiring leader
- Dr Arun Chandavarkar is the MD of Biocon Biologics
- Dr has been with Biocon since 1990
- Dr Chandavarkar holds Chemical Engineering from IIT and a Ph.D. in Biochemical Engineering from MIT
- Professionally managed board and team
Investors:
- Promoters hold 60.64% of the company
- Arohi Funds hold 3.89% of the company
Arohi is an absolute return investor and focuses on generating returns over the long run by investing in quality businesses run by trustworthy and competent people.
Arohi’s investments include Mindtree, Jubilant Food, Tube Investments, CG Power, Biocon.
Valuation:

Expect to have an IPO of Biocon Biologics in 18-24 months.
Resources:
- Annual Reports, Investor Presentations
Disclaimer:
For educational purposes only.
Appendix:
Branded drugs are either Synthetic or Biological.
Synthetic branded drugs are made from a chemical process.
It can be exactly replicated into more affordable generic versions.
Generic drugs are 40% to 50% cheaper than the brand product.
Biologics are drugs made out of living organisms like yeast, bacteria, or animal or plant cells.
A biosimilar is highly similar to the original biological drug.
Biologics involve large, complex molecules.
Biosimilars are 15% to 20% cheaper.
Biocon Biologics + Viatris:




Leave a comment